How to Balance:
HF + Ba(OH)2 → BaF2 + H2O
Word equation: Hydrofluoric acid + Barium hydroxide → Barium fluoride + water
Type of Chemical Reaction: For this reaction we have a neutralization reaction.
Balancing Strategies: This is a neutralization reaction since we have an acid, HF, and a base Ba(OH)2. That you could also consider it a double displacement some reaction.
Up and be sure to count all of the hydrogen atoms on the reactants side of the equation. People often have difficulty balancing these equations because they forget the hydrogen atom in the HF.
Essential Resources
For a complete explanation, watch:
When balancing chemical equations our goal is to have the same number of each type of atom on both sides of the equation.
Only change the coefficients (these are the numbers in front substances).
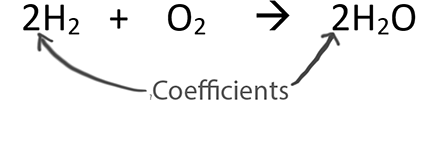
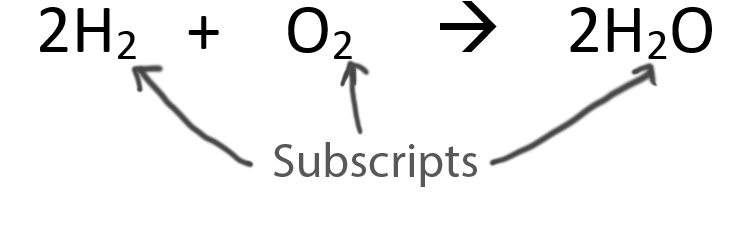
Video:
Subscribe