How to Balance:
CuSO4 + KI → CuI + I2 + K2SO4
Word equation: Copper (II) sulfate + Potassium iodide → Copper (I) iodide + Iodine gas + Potassium sulfate
Type of Chemical Reaction: For this reaction we have a double replacement (sort of) reaction.
Balancing Strategies: This is a little bit of a challenging reactions. I recommend trying to get an equal number of iodine atoms on the product side of the reaction. You can do this by changing the coefficient in front of the CuI.
You should end up with four iodine atoms on the product side of the equation.
Essential Resources
For a complete explanation, watch:
When balancing chemical equations our goal is to have the same number of each type of atom on both sides of the equation.
Only change the coefficients (these are the numbers in front substances).
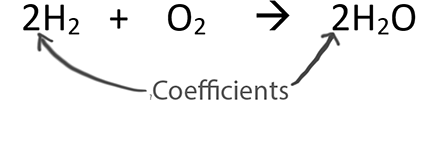
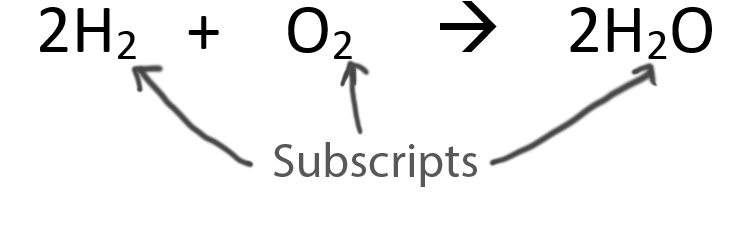
Video:
Subscribe