How to Balance:
Zn + H2O → Zn(OH)2 + H2
Word equation: Zinc + Water → Zinc hydroxide + Hydrogen gas
Type of Chemical Reaction: For this reaction we have a single displacement reaction.
Balancing Strategies: In this reaction of zinc plus water, we have a single displacement reaction. The reaction doesn't happen in cold water. The water has to be very hot or even steam.
The key to balancing this equation is correctly counting the number of hydrogen atoms on the product side of the equation.
Essential Resources
For a complete explanation, watch:
When balancing chemical equations our goal is to have the same number of each type of atom on both sides of the equation.
Only change the coefficients (these are the numbers in front substances).
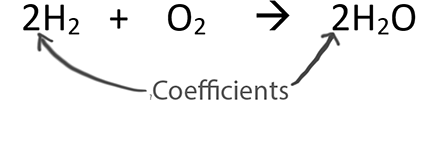
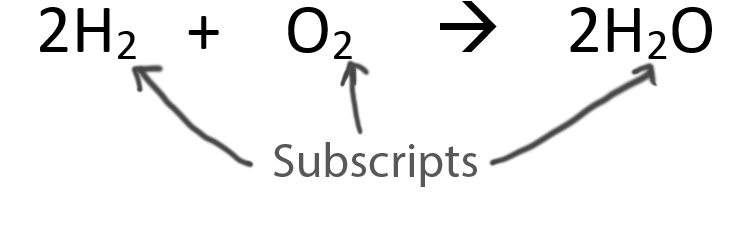
Video:
Subscribe