How to Balance:
Al2O3 + H2SO4 → Al2(SO4)3 + H2O
Word equation: Aluminum oxide + Sulfuric acid → Aluminum sulfate + Water
Type of Chemical Reaction: For this reaction we have a double replacement reaction.
Balancing Strategies: In this double replacement reaction we have Al2O3 + H2SO4 producing Al2(SO4)3 + H2O.
It is helpful to consider the SO4 as one item when balancing the equation. This will make things much easier.
Essential Resources
For a complete explanation, watch:
When balancing chemical equations our goal is to have the same number of each type of atom on both sides of the equation.
Only change the coefficients (these are the numbers in front substances).
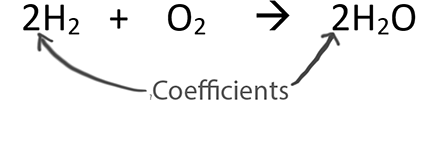
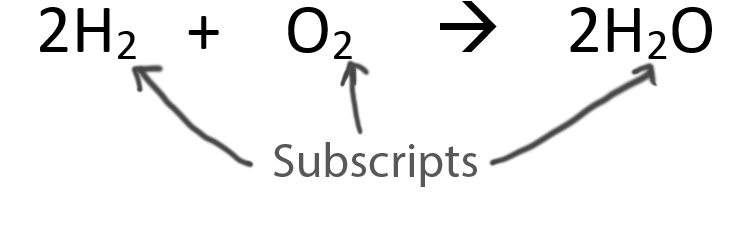
Video:
Subscribe