Equation Stoichiometry
Equation stoichiometry is the culmination of many skills:
- Calculating Molar Mass
- Converting from Moles to Grams, Liters, & Particles
- Balancing Chemical Equations
- Finding the Mole Ratio
- Estimating Amounts and Planning Strategies
We put all of these skills together to solve mass to mass, mass to liters, and mass to molecules stoichoimetry problems.
Solving Stoichiometry Problems:
In a typical problem we:
- convert grams, liters, or molecules given in the problem to moles.
- use the mole ratio to find moles of the desired quantity.
- convert moles of the desired quantity to grams, liters, or molecules. This is the answer!
More Help and Practice:
Thinking Tools
It is helpful to visualize two mole maps connected by the mole ratio to solve stoichiometry problems:
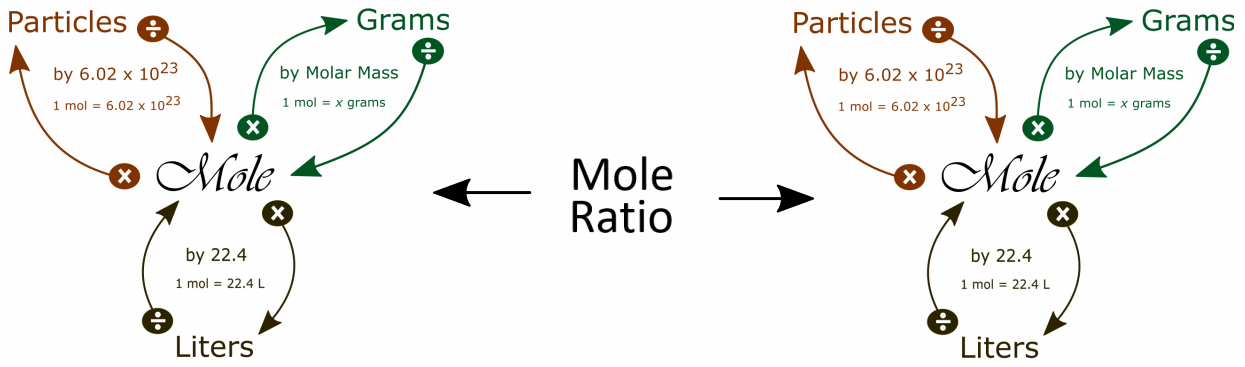
One way to find the the mole ratio is:

We get the coefficients for the mole ratio from the balanced equation.